The Drug Hypersensitivity and Desensitization Center serves patients with drug allergy, providing them with the opportunity to receive the first line treatments that are essential to their medical care. Through careful evaluation, select testing, and desensitization, the team of providers in the Center has facilitated the care of thousands of patients.
Mariana C. Castells, MD, Director, Drug Hypersensitivity and Desensitization Center at Brigham and Women's Hospital, describes the process of drug desensitization, a method in which a drug can be safely re-introduced to a patient who has become allergic to their medication. Read the Drug Desensitization video transcript.
| Stage | Description |
|---|---|
| Drug Reaction | An allergic reaction occurs after a patient is exposed to a medication |
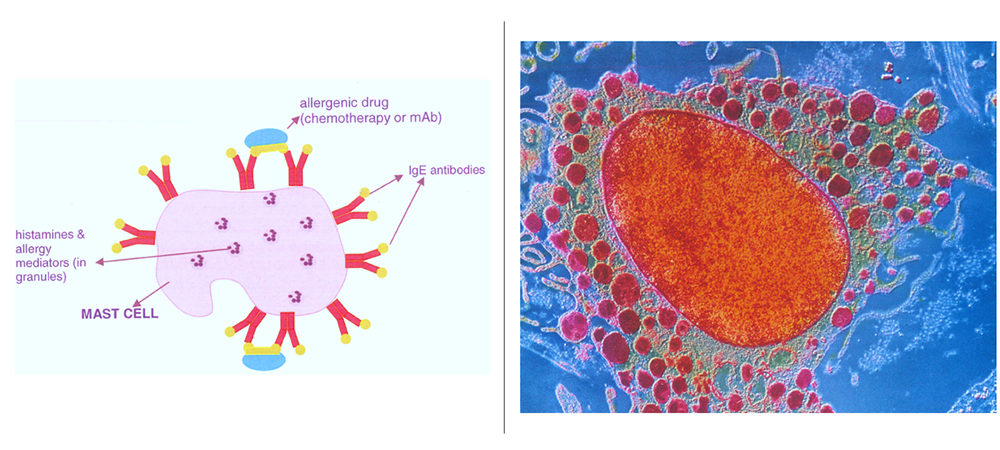
| Diagnostic Testing and Risk Stratification | A skin test and clinical evaluation shed insight into if a reaction was immunoglubluin-E (IgE) mediated or non-IgE mediated. IgE is the type of antibody that maps specificity to an allergy. For example, if a patient reacts to their first-line chemotherapy or other medication they probably have an IgE-specific allergy to the drug circulating in the blood |
| Protocol | The prescribed dose of the medication is placed into an algorithm to create a 1 bag / 4 step, 2 bag / 8 step or 3 bag / 12 step infusion protocol. Decisions on protocols are made by an allergist and are based on the mechanism of the reaction, skin testing and patient symptoms |
| Treatment | The treatment introduces small increasing doses of drug to the patient in 15-minute intervals until the patient is considered desensitized and the final dose is safely reached. This occurs on one day over several hours. |
| Tolerance | The desensitized state is temporary and only lasts for a short window of time. Once a patient enters the desensitization program, all subsequent infusions of the allergic drug will be given as desensitizations for as long as needed or until an allergist deems it safe to receive the allergic drug in normal infusion |
Director
Associate Director
Attending Physicians
Desensitization Nurse Practitioner
Desensitization Pharmacist
Desensitization Coordinator
For providers : Please contact Kylie Besz, Desensitization Coordinator, at 617-5251222, if interested in having your patient evaluated for possible desensitization.
For patients : Please contact your doctor to evaluate the possibility of desensitization. They will need to contact the coordinator directly
All patients must be referred by a physician. Referral Forms must be filled out by a referring physician.
Brigham and Women’s Hospital
60 Fenwood Road
Hale Building for Transformative Medicine
Boston, MA 02115
Phone: (617) 732-9850
For over a century, a leader in patient care, medical education and research, with expertise in virtually every specialty of medicine and surgery.
About BWH