TMS is a non-invasive brain stimulation treatment for patients whose psychiatric or neurological symptoms have not improved with routine treatments like medications and therapy. The United States Food and Drug Administration (FDA) has cleared TMS for the following indications:
The FDA has also granted TMS a “breakthrough device designation” for bipolar depression, but it has not yet cleared it for this indication. Nearly all insurance companies cover TMS for depression, but they may or may not cover TMS for other indications. Please contact your insurance directly for further information. This page will focus on treatment-resistant depression, the illness most commonly treated with TMS.
Depression is among the leading causes of disability worldwide. Two out of three people taking antidepressant medications continue to experience symptoms of depression. Moreover, the probability of getting well and staying well decreases each time a patient tries a new medication. TMS is a safe and effective treatment that can work even after psychotherapy and medications have not worked. Many patients consider TMS before other FDA-approved or cleared treatments for depression such as esketamine (brand name Spravato) or electroconvulsive therapy (ECT) because TMS does not require anesthesia, medication, or invasive procedures. TMS is usually well tolerated, and patients can drive themselves to and from treatment.
TMS uses an electromagnetic coil to create magnetic pulses (similar to an MRI), which pass through the skull to stimulate the brain network involved in regulating mood. As such, it is generally not recommended for patients with seizure disorders or patients who have metal in the head area. Daily (weekday) treatment sessions for 6-8 weeks (30-36 treatments total) are typically required.
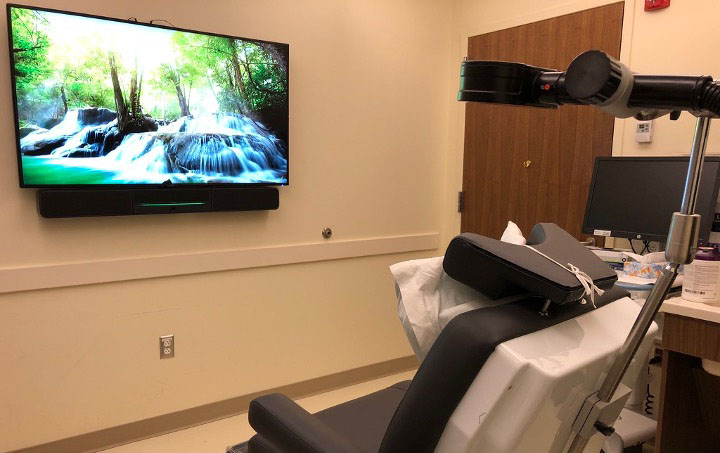
During treatment sessions, patients recline in a chair while a TMS coil rests on the specific part of their head. The TMS coil makes a clicking sound and produces a tapping sensation when it is stimulating the brain. Patients are awake for the entirety of the session, which ranges from 3 to 37 minutes, depending on the treatment protocol prescribed. Studies have shown that the antidepressant effects of TMS last after the acute series of treatments ends.
Common
Uncommon
Most insurance plans cover TMS after a patient does not benefit from or cannot tolerate 3-4 antidepressant medications.
We are optimized to reduce exposure to the virus that causes COVID-19 and offer:
Our physician scientists specialize in discovering and testing new treatment targets for brain stimulation therapies like TMS. We offer observational trials that do not affect routine TMS treatment as well as clinical trials that test new treatment strategies. If interested, please e-mail bwhtmstrials@bwh.harvard.edu or check clinicaltrials.gov.
All patients must have a referring clinician, someone who follows them longitudinally for psychiatric care. Referral methods are listed below:
For more information:
For over a century, a leader in patient care, medical education and research, with expertise in virtually every specialty of medicine and surgery.
About BWH